Radioactive wastes are generated as a consequence of numerous processes and operations. These range from the reprocessing of spent nuclear fuel and plutonium production for weapon applications, to mining and refining of uranium ore, commercial research activities and use of isotopes, and medical, hospital and university activities. Unprocessed spent nuclear fuel itself has also been considered as a waste. Radioactive waste management practices vary worldwide but share the common interest of treating these wastes as highly hazardous materials from which the environment must be protected.
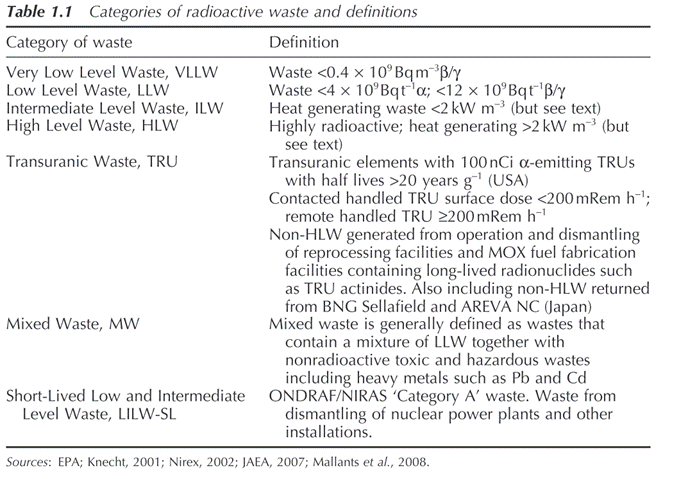
Radioactive wastes are generally subdivided into a number of specific categories depending on their overall activity, as summarized in Table 1.1 . These include very low level waste (VLLW), low level waste (LLW), intermediate level waste (ILW) and high level waste (HLW).
The potential use of glass as a nuclear wasteform was initially investigated in the early 1950s in Canada using a natural silicate material, nepheline syenite, as the starting mate rial. This was mixed with an acidic solution of the waste together with lime, and the mixture melted at 1250 – 1350 ° C in a fire – clay crucible. In the late 1970s and early 1980s, the UK and USA to vitrified nuclear wastes using borosilicate glass. Despite an apparently strong technological background, and as noted by Plodinec (2000) , the development of suitable HLW glass compositions was not straightforward or undemanding. Commercial glass technology is concerned with the production of glasses of specific, well – defined compositions, which give rise to glasses of known and reproducible properties. HLW glasses, however, due to the inevitable variability of wastestream compositions, give rise to vitrified products that are not consistent in their properties.
Suitable HLW glass compositions must therefore be very tolerant of compositional variations, whilst still providing a product with acceptable properties and meeting quality control requirements. They also contain constituents rarely found in common commercial glasses, together with much higher concentrations of common minor commercial glass constituents like Fe, Ni and Mn. Their resultant properties are therefore open to further uncertainty. In addition, unlike their commercial counterparts, normal maintenance of HLW vitrification plants can only be carried out remotely, adding to the complexity, running difficulties, and cost of such plants.
The Nano-Photonic glass Laboratory is researching the technology about the nuclear waste vitrification.
